Virginia Tech researcher shines light on origin of bioluminescence
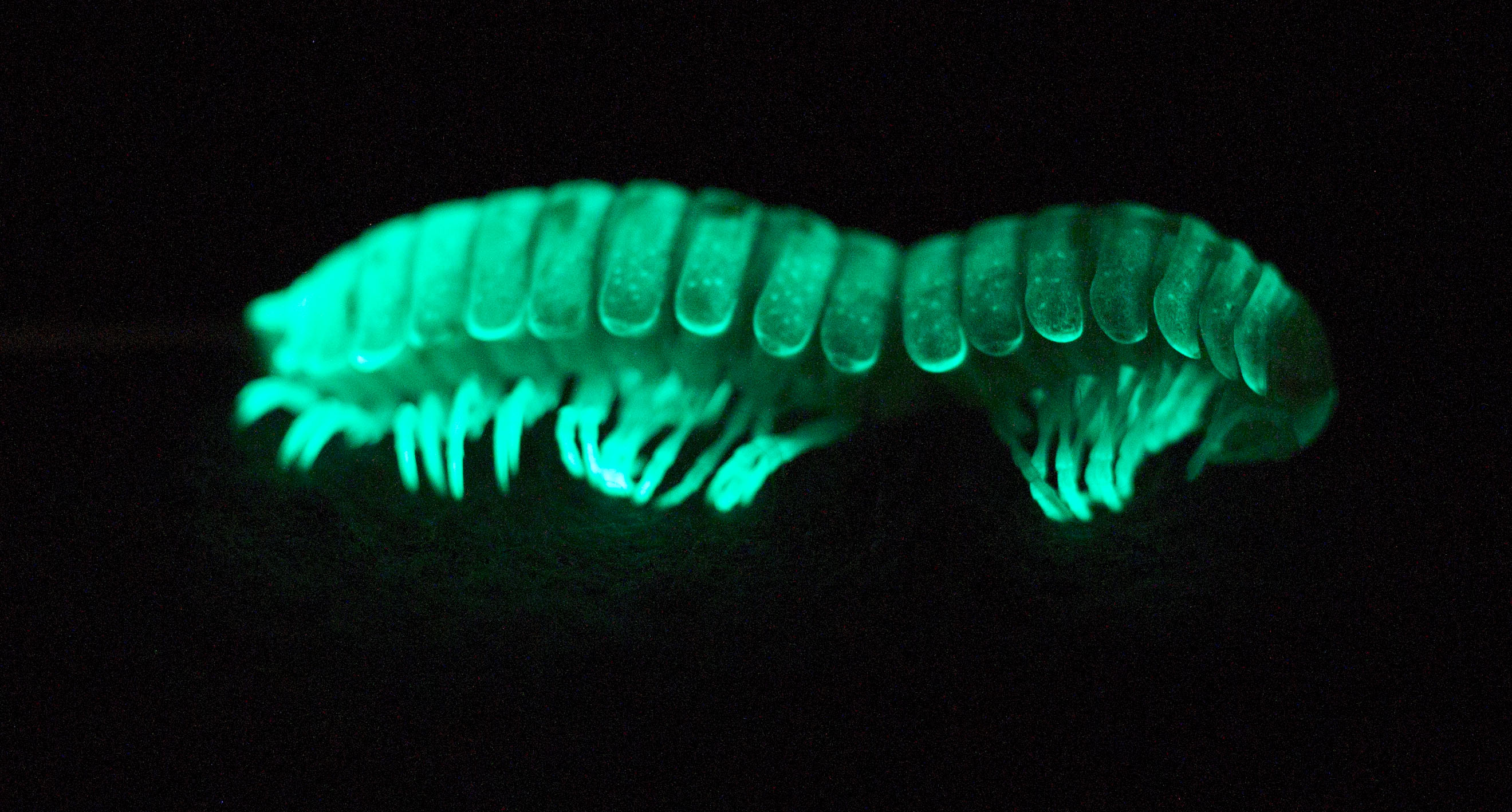
A Virginia Tech scientist has discovered that bioluminescence may not have originated as a means to ward off predators, but instead evolved as a way to survive in harsh climates — at least in one millipede.
The finding, based on the discovery of a millipede that hadn’t been seen in 50 years, shows that even the seemingly most complex and intricate of traits can be traced in evolution as small steps leading to a complex feature we see today.
The results of the evolutionary mapping of the bug were published Monday in the Proceedings of the National Academy of Sciences.
“Living things glow in many different colors and for many different reasons, but now we know that the early evolutionary role of bioluminescence may be completely different than its modern day function,” said Paul Marek, an assistant professor of entomology in the College of Agriculture and Life Sciences. “This discovery clarifies the evolutionary origins of many complex traits, not just bioluminescence.”
Marek began studying bioluminescence in millipedes seven years ago and the feature always seemed difficult to understand and trace in terms of its early origins.
“Because we’re looking at a trait that has been shaped by millions of years of evolution these traits often bear little resemblance to their ancestral form,” said Marek.
But Marek rediscovered a significant piece of the luminosity puzzle while revisiting a Californian millipede that had been literally shelved for half a century. The bug, Xystocheir bistipita, had been collected in 1967 and was not seen again until Marek rediscovered it during fieldwork in the foothills of San Luis Obispo, California.
Diminutive in size compared to other millipedes, the species lives at a lower elevation with few predators and was not thought to belong to the genus of millipedes that glow called Motyxia — the only genus of bioluminescent millipedes in the Western Hemisphere.
After sequencing the bug’s DNA, he found that it was in fact related to its luminous cousins, and changed its name to Motyxia bistipita.
Unlike fireflies that glow because of an enzymatic reaction between a luciferase and a luciferin, M. bistipita owes its soft green-blue glow to the reaction of a photoprotein that requires magnesium and is thought to have initially evolved for its antioxidant properties to cope with the oxidative stress of living in a low-lying, dry environment. Bioluminescence was then repackaged as a nocturnal warning signal in millipedes that live at a higher elevation and contend with many more predators.
“After we sequenced them we were able to place the millipede on an evolutionary tree with other bioluminescent species in Motyxia,” said Marek. “We demonstrated the faint bioluminescence of the low-lying millipedes represented an older trait and the brighter luminescence of their mountain cousins represented a newer trait.”
Marek mapped the brightness of light in M. bistipita, and its close relatives that live at higher elevation and glow more brightly, and demonstrated a gradation of brightness from faint to bright.
To reconstruct the evolutionary tree, Marek and his team included every species in the genus Motyxia and all its closest nonglowing relatives and tested the relationship between them and M. bistipita. Marek and his team measured light emission photographically in a darkroom by placing individual millipedes in a light tube. The researchers also measured toxicity of the bugs by measuring the volume of cyanide glands that line the sides of the millipedes. Bugs with larger cyanide glands were also brighter, indicating a functional link between luminescent intensity of displays and toxicity.
“Evolution of a complex feature such as bioluminescence is difficult to understand at first glance but totally makes sense when considered in an evolutionary context,” said Marek. “We showed that bioluminescence, a trait typically used as a warning pattern evolved gradually and for a different purpose.”
Marek’s research is funded by the National Science Foundation and National Geographic Society.





