Researchers discover compounds that could decrease fungal infection in lungs
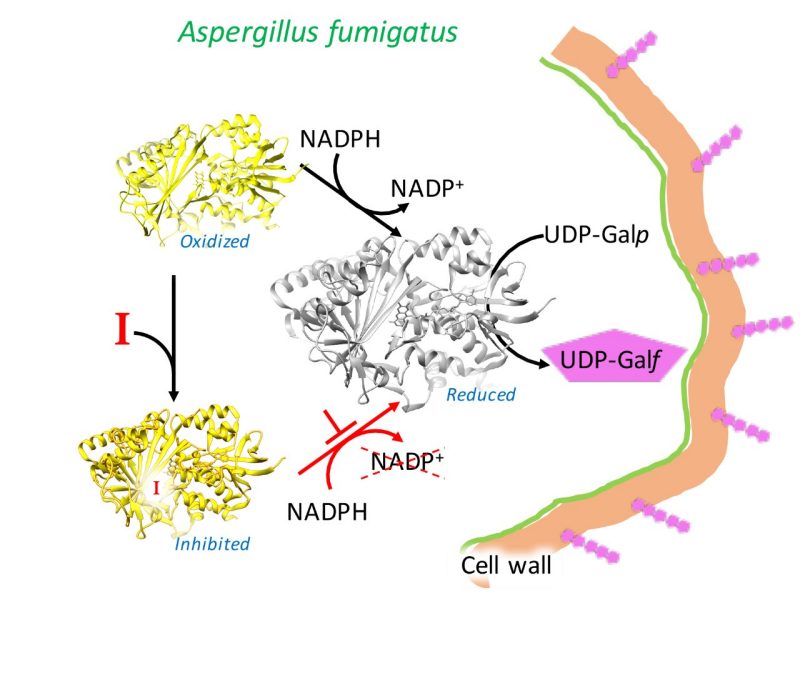
A Virginia Tech research team found a way to alter the growth of a fungus that causes deadly lung infections in immune-compromised individuals, such as leukemia patients and recipients of stem cell or organ transplants.
Two naturally occurring compounds—identified in the small molecule library available at the Virginia Tech Center for Drug Discovery Screening Laboratory supported by the Fralin Life Science Institute — work against an enzyme important in cell wall production in the deadly fungus, called Aspergillus fumigatus.
The findings were published in the journal Scientific Reports on Sept. 7.
“This work validates the use of an assay designed to target a specific region of UGMs. The compounds that were identified are the first generation inhibitors of UGM from eukaryotic human pathogens,” said Pablo Sobrado, lead author, professor of biochemistry in the College of Agriculture and Life Sciences, and a Fralin Life Science Institute affiliate.
The compounds, naringenin and hesperetin, are flavonones found in citrus fruits. Naringerin has been shown to have antibacterial activity and hesperetin is known to be an anti-inflammatory and to help with regulatory adjustment of the immune system.
The enzyme, called UDP-galactopyranose mutase, produces a rare sugar that is a major component of the cell wall in several dangerous organisms besides Aspergillus fumigatus, such as other fungi, parasitic worms, and protozoan parasites. It is also known to help the bacteria that causes tuberculosis to infect its human hosts.
In 2010, Sobrado received funding from the National Institutes of Health to study UDP-galactopyranose mutases. The overall goal in studying enzyme structure and function is to develop improved treatments for human disease.
The next step will be to fully describe the structure of the enzyme with the inhibitor bound, which will help determine how it works. These structures will provide a blueprint for the development of more effective derivatives. The team will also search for more potent compounds that can be developed into potential drugs to treat fungal-related infections and other diseases.
Co-authors on the paper include Julia S. Martin del Campo, a postdoctoral research scientist in Sobrado’s lab; Meital Eckshtain-Levin, a postdoctoral research scientist in Sobrado’s lab; and Nancy Vogelaar, manager of the Virginia Tech Center for Drug Discovery.







