Study takes a unique approach to new generation of smart drug delivery carriers

Imagine a tiny capsule, smaller than the tip of a needle, that could be programmed to release medicine at a specific location in your body and is inexpensive, easy to make, and more effective than the traditional pharmaceuticals we know today.
In addition, after delivering its medical contents, the capsule disappears because it’s biodegradable and made up of microscopic elements that are safely absorbed into the bloodstream.
Such a method of drug delivery would usher in a new era of pharmaceuticals. Some of the world’s most devastating illnesses could be better treated with lifesaving drugs that are cheaper, more widely accessible, and perform better in the human body.
A Virginia Tech research team is one step closer to realizing that long-term vision. Biomedical engineering and mechanics and mechanical engineering faculty and students have spent the past year testing the viability of using a unique class of designed materials, coupled with a surprising trigger, to build smarter drug delivery systems.
Their research, published in RSC Advances, outlines a proof of concept for using focused ultrasound waves to activate shape-memory polymers. Most recently gaining attention for their use in designing biocompatible devices, shape-memory polymers can be used to deliver drugs inside the human body.
Aarushi Bhargava, a second-year Ph.D. student in Virginia Tech’s engineering mechanics program and the study’s lead author, described the research as an important first step in using shape-memory polymers to design and optimize efficient drug delivery systems for human use.
“With the help of ultrasound, these systems can deliver drugs in a controlled manner at the desired target location over a prolonged period of time, something that has been very difficult to do in the field of drug delivery mechanisms,” said Bhargava. “Shape-memory polymers give us an advantage because they’re flexible, biodegradable, and cost-effective. They’re also easy to manufacture.”
Shape-memory polymers are a class of smart materials that have the ability to return from a deformed, temporary shape to their original permanent shape when subjected to an external stimulus, such as light or heat.
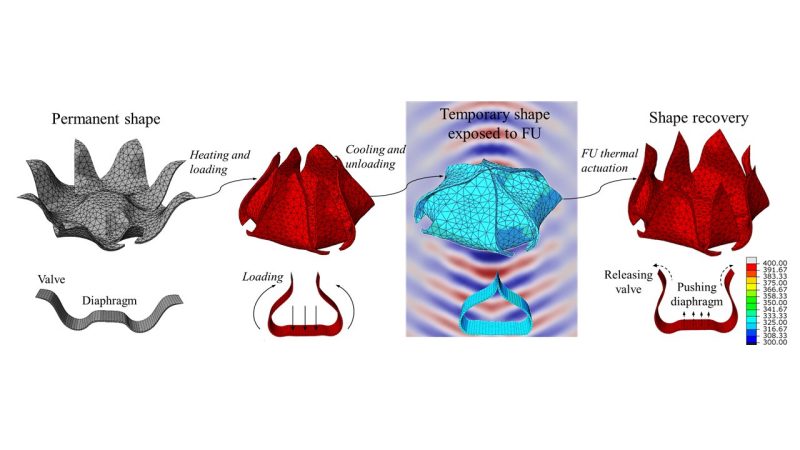
In this project, a conceptual framework for designing a shape-memory polymer container is loaded with drug particles in its original shape, heated, and deformed to its temporary shape. This temporary shape effectively packages the drug particles inside a tiny capsule-like container. When the capsule reaches its desired location within the body, it undergoes shape recovery through exposure to focused ultrasound and releases the loaded drug particles.
Focused ultrasound waves are those with frequencies higher than the upper audible limit of human hearing. The use of this unusual trigger to activate the shape-memory polymers is what sets the team’s findings apart from others doing similar work in the field of drug delivery systems.
The benefits of using focused ultrasound waves to activate the shape-memory polymer drug delivery container, instead of light or heat, include the flexible, noninvasive nature of the stimulus. Previous shape-memory polymer containers have relied on natural body heat for activation and can be difficult to control. Other noninvasive methods, such as magnetic fields or exposure to light, require special particles to generate a response. These additional particles can compromise the biodegradability and biocompatibility of shape-memory polymers.
Shima Shahab, an assistant professor in the Department of Biomedical Engineering and Mechanics and Bhargava’s faculty advisor, co-authored the study alongside Reza Mirzaeifar, an assistant professor of mechanical engineering; Jerry Stieg, a mechanical engineering undergraduate; and Kaiyuan Peng, a Ph.D. student in the mechanical engineering program, all of Virginia Tech.
Shahab explained the study’s findings would pave the way for designing more-efficient drug delivery capsules in the future, especially those that can be activated by focused ultrasound waves.
“We developed an important experimental-computational framework that can be used to design various ultrasound-activated drug delivery containers,” said Shahab. “The frameworks in this study can be specifically tailored for different applications depending on the size of drug particles, target time for releasing the particles, and the size and shape of the container.”
In addition to garnering attention in the field of drug delivery systems, the findings recently won the award for Best Student Paper at the 2017 Conference on Smart Materials, Adaptive Structures, and Intelligent Systems in Snowbird, Utah.
Shahab and Mirzaeifar first designed the original project in a collaboration between Virginia Tech’s MInDS and MultiSMArt labs in August 2016. While the team’s methods are still years away from clinical testing in humans, they have established an important foundation for future research.
“The study’s outcome moves us one step closer to the introduction of an efficient new generation of drug delivery systems,” said Mirzaeifar. “Our research will continue to focus on this goal.”
Written by Emily Roediger







